Esfenvalerate
 | |
| Names | |
|---|---|
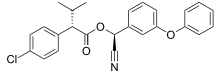
| Preferred IUPAC name (S)-Cyano(3-phenoxyphenyl)methyl (2S)-2-(4-chlorophenyl)-3-methylbutanoate | |
| Other names Asana (S)-Fenvalerate | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
Beilstein Reference | 4275674 |
| ChEBI |
|
| ChEMBL |
|
| ChemSpider |
|
| ECHA InfoCard | 100.118.804 |
| EC Number |
|
| KEGG |
|
PubChem CID |
|
| UNII |
|
| UN number | 3349 |
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | C25H22ClNO3 |
| Molar mass | 419.91 g·mol−1 |
| Density | 1.211 g/cm3 |
| Melting point | 60 °C (140 °F; 333 K) |
| log P | 6.22 |
| Vapor pressure | 0 mmHg at 25 °C |
| Hazards | |
| GHS labelling: | |
Pictograms |     |
| Danger | |
| H301, H313, H316, H317, H320, H330, H335, H370, H373, H410 | |
| P260, P261, P264, P270, P271, P272, P273, P280, P284, P301+P310, P302+P352, P304+P340, P305+P351+P338, P307+P311, P310, P312, P314, P320, P321, P330, P332+P313, P333+P313, P337+P313, P363, P391, P403+P233, P405, P501 | |
| Flash point | 256 °C (493 °F; 529 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references | |
Esfenvalerate is a synthetic pyrethroid insecticide marketed under the brand Asana.[2] It is the (S)-enantiomer of fenvalerate.[3]
In the United States, a limit of .05 ppm of the chemical's residue is permissible in food.[4]
References
- ^ Kelly, Kevin. "Environmental Fate of Esfenvalerate". California Environmental Protection Agency. Retrieved January 10, 2013.
- ^ Fishel, Frederick M. (2012). "Pesticide Toxicity Profile: Synthetic Pyrethroid Pesticides". University of Florida. Archived from the original on May 12, 2016. Retrieved January 10, 2013.
- ^ "Esfenvalerate". EXTONET (Extension Toxicology Network). Cooperative Extension Offices of Cornell University, Michigan State University, Oregon State University, and University of California at Davis. May 1994. Retrieved January 10, 2013.
- ^ The Code of Federal Regulations of the United States of America. U.S. Government Printing Office. 2006. pp. 445–446.
- v
- t
- e
- Aluminium phosphide
- Boric acid
- Chromated copper arsenate
- Copper(II) arsenate
- Copper(I) cyanide
- Cryolite
- Diatomaceous earth
- Lead hydrogen arsenate
- Paris Green
- Scheele's Green
- Acephate
- Azamethiphos
- Azinphos-methyl
- Bensulide
- Chlorethoxyfos
- Chlorfenvinphos
- Chlorpyrifos
- Chlorpyrifos-methyl
- Coumaphos
- Demeton-S-methyl
- Diazinon
- Dichlorvos
- Dicrotophos
- Diisopropyl fluorophosphate
- Dimefox
- Dimethoate
- Dioxathion
- Disulfoton
- Ethion
- Ethoprop
- Fenamiphos
- Fenitrothion
- Fenthion
- Fosthiazate
- Isoxathion
- Malathion
- Methamidophos
- Methidathion
- Mevinphos
- Mipafox
- Monocrotophos
- Naled
- Omethoate
- Oxydemeton-methyl
- Parathion
- Parathion-methyl
- Phenthoate
- Phorate
- Phosalone
- Phosmet
- Phoxim
- Pirimiphos-methyl
- Quinalphos
- R-16661
- Schradan
- Temefos
- Tebupirimfos
- Terbufos
- Tetrachlorvinphos
- Tribufos
- Trichlorfon
- Acrinathrin
- Allethrins
- Bifenthrin
- Bioallethrin
- Cyfluthrin
- Cyhalothrin
- Cypermethrin
- Cyphenothrin
- Deltamethrin
- Empenthrin
- Esfenvalerate
- Etofenprox
- Fenpropathrin
- Fenvalerate
- Flumethrin
- Fluvalinate
- Imiprothrin
- Metofluthrin
- Permethrin
- Phenothrin
- Prallethrin
- Pyrethrin (I, II; chrysanthemic acid)
- Pyrethrum
- Resmethrin
- Silafluofen
- Tefluthrin
- Tetramethrin
- Tralomethrin
- Transfluthrin
- Afoxolaner
- Amitraz
- Azadirachtin
- Bensultap
- Buprofezin
- Cartap
- Chlordimeform
- Chlorfenapyr
- Cyromazine
- Fenazaquin
- Fenoxycarb
- Fipronil
- Fluralaner
- Hydramethylnon
- Indoxacarb
- Limonene
- Lotilaner (+milbemycin oxime)
- Pyridaben
- Pyriprole
- Sarolaner
- Adjuvants (Piperonyl butoxide, Sesamex)
- Spinosad
- Sulfluramid
- Tebufenozide
- Tebufenpyrad
- Veracevine
- Xanthone
- Metaflumizone
 | This article about an organic compound is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e












