Pelargonidin
 | |
| Names | |
|---|---|
| IUPAC name 3,4′,5,7-Tetrahydroxyflavylium | |
| Systematic IUPAC name 3,5,7-Trihydroxy-2-(4-hydroxyphenyl)-1λ4-benzopyran-4-ylium | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChEBI |
|
| ChEMBL |
|
| ChemSpider |
|
| KEGG |
|
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | C15H11O5+ |
| Molar mass | 271.24 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  N verify (what is N verify (what is  Y Y N ?) N ?) Infobox references | |
Pelargonidin is an anthocyanidin, a type of plant pigment producing a characteristic orange color used in food and industrial dyes.[1]
Natural occurrences
Presence in flowers
Pelargonidin can be found in red geraniums (Geraniaceae). It is the predominant pigment causing the red coloration in the spathes of Philodendron (Araceae). The orange-coloured flowers of blue pimpernel (Anagallis monelli, Myrsinaceae) have a higher concentration of pelargonidin pigment. Red and Pink Roses (Rosa) obtain their color from this phytochemical. [2]
Presence in food
Pelargonidin can be found in berries such as ripe raspberries and strawberries, as well as blueberries, blackberries, cranberries but also in saskatoon berries[3] and chokeberries. It is also found in plums and pomegranates. Pelargonidin gives red radishes their color.[4]
It is present in large amounts in kidney beans.[5]
Glycosides
In many plant systems, Pelargonidin can be added to a glucose molecule to form Pelargonidin 3-glucoside (callistephin). This is done by the 3GT, anthocyanin 3-O-glucosyltransferase gene. [6]
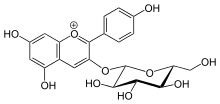
However this glucosidation reduces its antioxidant activity,[7] and changes the wavelength of max light absorbance from 520 nm to 516 nm. [8]
Acylated pelargonidin glycosides can be found in red-purple flowers of Ipomoea purpurea.[9]
See also
References
- ^ US granted 6,767,999, Smirnov, Vitaly; Sidorov, Viktor; Smirnova, Valentina, "Anthocyantin coloring agent and method for the production thereof from organic matter", published Nov 01, 2001, issued July 27, 2004
- ^ Huihua, Wan; Chao, Yu; Yu, Han; Xuelian, Guo (2019). "Determination of Flavonoids and Carotenoids and Their Contributions to Various Colors of Rose Cultivars (Rosa spp.)". Frontiers in Plant Science. 10: 123. doi:10.3389/fpls.2019.00123. PMC 6379320. PMID 30809238.
- ^ Mazza, G. (2005). "Compositional and Functional Properties of Saskatoon Berry and Blueberry". International Journal of Fruit Science. 5 (3): 101–120. doi:10.1300/J492v05n03_10. S2CID 85691882.
- ^ Takeshi Nishio (4 October 2017). Takeshi Nishio, Hiroyasu Kitashiba (ed.). The Radish Genome. Springer. p. 4. ISBN 978-3-319-59253-4.
- ^ Lin, Long-Ze; Harnly, James M.; Pastor-Corrales, Marcial S.; Luthria, Devanand L. (2008). "The polyphenolic profiles of common bean (Phaseolus vulgaris L.)". Food Chemistry. 107 (1): 399–410. doi:10.1016/j.foodchem.2007.08.038. PMC 4276374. PMID 25544796.
- ^ Levisson, Mark; Patinios, Constantinios; Hein, Sascha; de Groot, Phillip A. (2018). "Engineering de novo anthocyanin production in Saccharomyces cerevisiae". Microbial Cell Factories. 17 (103): 103. doi:10.1186/s12934-018-0951-6. PMC 6029064. PMID 29970082.
- ^ Li, Wenfeng; Gu, Mengyuan; Gong, Pengling; Wang, Jinxia (2021). "Glycosides changed the stability and antioxidant activity of pelargonidin". Lebensmittel-Wissenschaft & Technologie. 147 (3): 111581. doi:10.1016/j.lwt.2021.111581. S2CID 235531625.
- ^ Gould, Kevin S. (2009). Anthocyanidins: Biosynthesis, Functions, and Applications. New York: Springer. p. 286. ISBN 978-0-387-77334-6.
- ^ Saito, N; Tatsuzawa, F; Yokoi, M; Kasahara, K; Iida, S; Shigihara, A; Honda, T (1996). "Acylated pelargonidin glycosides in red-purple flowers of Ipomoea purpurea". Phytochemistry. 43 (6): 1365–70. doi:10.1016/s0031-9422(96)00501-8. PMID 8987912.
External links

- v
- t
- e
- 5-Desoxy-peonidin
- Aurantinidin
- Cyanidin
- 6-Hydroxycyanidin
- Delphinidin
- Fisetinidin
- Guibourtinidin
- Pelargonidin
- Robinetinidin
- 5-Desoxy-malvidin
- Capensinidin
- Europinidin
- Hirsutidin
- Kaempferidinidin
- Malvidin
- Peonidin
- Petunidin
- Pulchellidin
- Rosinidin
(anthocyaninidin glycosides)
- Callistephin (Pelargonidin 3-O-glucoside)
- Chrysanthemin (Cyanidin 3-O-glucoside)
- Myrtillin (Delphinidin 3-O-glucoside)
- Oenin (Malvidin 3-O-glucoside)
- Peonidin 3-O-glucoside
- Petunidin 3-O-glucoside
- Pulchellidin 3-glucoside
Diglucosides:
- Cyanin (Cyanidin 3,5-O-diglucoside)
- Delphin (Delphinidin 3,5-O-diglucoside)
- Malvin (Malvidin 3,5-diglucoside)
- Pelargonin (Pelargonidin 3,5-O-diglucoside)
- Peonin (Peonidin 3,5-O-diglucoside)
- Petunin (Petunidin 3,5-O-diglucoside)
Others glycosides:
- Antirrhinin (Cyanidin 3-O-rutinoside)
- Ideain (Cyanidin 3-O-galactoside)
- Delphinidin 3-O-rhamnoside
- Petunidin 3-O-arabinoside
- Petunidin 3-O-galactoside
- Petunidin 3-O-rhamnoside
- Petunidin 3-O-rutinoside
- Primulin (Malvidin 3-O-galactoside)
- Pulchellidin 3-rhamnoside
- Tulipanin (Delphinidin 3-O-rutinoside)
| Acetylated anthocyanins | |
|---|---|
| Coumaroylated anthocyanins (cis- and trans-) |
|
| Caffeoylated anthocyanins |
|
| Malonylated anthocyanins |
|
| Acylated anthocyanin diglycosides |
|
- Malvidin glucoside-ethyl-catechin
- Catechin(4α→8)pelargonidin 3-O-β-glucopyranoside
- Epicatechin(4α→8)pelargonidin 3-O-β-glucopyranoside
- Afzelechin(4α→8)pelargonidin 3-O-β-glucopyranoside
- Epiafzelechin(4α→8)pelargonidin 3-O-β-glucopyranoside
- Metalloanthocyanins (commelinin)
- Cyanosalvianin
- Protocyanin
- Protodelphin)
- Pyranoanthocyanins
- Copigmentation
- Anthocyanone A (degradation product of oenin)
- Malvone (oxidation product of malvin)










