Apicidin
 | |
| Names | |
|---|---|
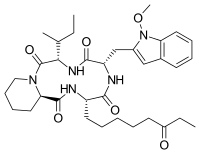
| Preferred IUPAC name (3S,6S,9S,15aR)-9-[(2S)-Butan-2-yl]-6-[(1-methoxy-1H-indol-3-yl)methyl]-3-(6-oxooctyl)octahydro-2H-pyrido[1,2-a][1,4,7,10]tetraazacyclododecine-1,4,7,10(3H,12H)-tetrone | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.163.614 |
IUPHAR/BPS |
|
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | C34H49N5O6 |
| Molar mass | 623.795 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references | |
Chemical compound
Apicidin is a fungal metabolite, as well as a histone deacetylase inhibitor.[1]
References
- ^ Han, J. W.; Ahn, S. H.; Park, S. H.; Wang, S. Y.; Bae, G. U.; Seo, D. W.; Kwon, H. K.; Hong, S.; Lee, H. Y.; Lee, Y. W.; Lee, H. W. (November 2000). "Apicidin, a histone deacetylase inhibitor, inhibits proliferation of tumor cells via induction of p21WAF1/Cip1 and gelsolin". Cancer Res. 60 (21): 6068–74. PMID 11085529.
- v
- t
- e
Histone deacetylase inhibitors
- 3,3'-Diindolylmethane
- β-Hydroxybutyric acid (β-hydroxybutyrate)
- Abexinostat
- Acetoacetic acid (acetoacetate)
- Allyl mercaptan
- Apicidin
- Belinostat
- Butyric acid (butyrate)
- Capsaicin
- Citarinostat
- Curcumin
- Diallyl disulfide
- Entinostat
- Fimepinostat
- Givinostat
- Indole-3-carbinol
- Kevetrin
- Martinostat
- Mocetinostat
- Niacinamide
- Panobinostat
- Parthenolide
- Phenylbutyrate
- Pracinostat
- Quisinostat
- Resminostat
- Romidepsin
- Scriptaid
- Sodium butyrate
- Sodium oxybate (GHB sodium)
- Sodium phenylbutyrate
- Sodium valproate
- Sulforaphane
- Trapoxin B
- Trichostatin A
- Tucidinostat
- Valnoctamide
- Valproic acid (valproate)
- Valproate pivoxil
- Valproate semisodium
- Valpromide
- Vorinostat (SAHA)
See also: Receptor/signaling modulators
 | This article about an organic compound is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e











